Colligo Academy Pocus Basics
Innehållsförteckning
Cardiac Ultrasound
While traditional physical examination and hemodynamic monitoring (both invasive and non-invasive) often provide extremely valuable information, assessment of cardiac structures by ultrasound can rapidly provide data that would be otherwise inapparent. Items of particular interest to the clinician at the bedside include the assessment of left ventricle (LV) function, markers of the patient’s volume status, and the presence or absence of pericardial effusion and/or tamponade physiology.
Point of Care Cardiac Ultrasound Applications in the Emergency Department and Intensive Care Unit - A ReviewRobert T Arntfield & Scott J Miliington, 2012
Objectives
To reach the objectives for this module read the following text, watch the videos and finish the QUIZ.
- Acquiring decent images with ultrasound of the four main cardiac windows (subcostal, PLAX, PSAX, and apical 4 chamber view) and being able to identify relevant strucures
- The ability to identify and assess the FATE (Focus Assessed Transthoracic Echo) questions:*
- Look for obvious pathology
- Assess wall thickness + chamber dimensions
- Assess bi-ventricular function
- Image pleura on both sides
- Relate the information to the clinical context
- Apply additional ultrasound
- Understanding the pitfalls and difficulties associated with bedside cardiac ultrasound
1. Introduction
Bedside point-of-care cardiac ultrasound is a focused assessment that for the starting practitioner will help answer two main questions quickly and with relatively little practice:
- Is there pericardial fluid present?
- Is the heart beating?
In the emergency room setting those two questions can be critical to answer and the skills required to do just that can be acquired relatively quickly too (as in the scope for a course like this one). For this course, however, we have also included the six FATE assessment points mentioned above. BUT, bear in mind that cardiac ultrasound is difficult and takes more time and practice than the other modules if you want to progress beyond the two main focused questions.
Cardiac ultrasound is the most difficult of the selected bedside ultrasound exams in this course to become proficient in. Skilled echocardiographers spend years becoming good at cardiac ultrasound, and at Karolinska our cardiology residents must spend at least 6 months of their residency at their echocardiography rotation. For a short “crash course” in POCUS like this, the primary goal is therefore only to present you to bedside cardiology and the learning objective to be more sufficient in judging the two primary focused questions mentioned above. The FATE questions should therefore be seen as only an introduction to extended evaluation of cardiac ultrasound, as there is no possibility to become proficient in the skillset needed to become sufficiently confident (and accurate) during this course. However, we expect you to apply and practice bedside-ultrasound after the course and it’s in your daily clinical work where you will become more proficient in judging and evaluating the six FATE objectives mentioned above.
2. The Four Cardiac Windows and Orientation
There are generally four main cardiac-windows that every POCUS-practioner should know and be able to acquire with the cardiac probe after a basic POCUS course. If you have never practiced cardiac ultrasound previously, my advise would be to just accept the different windows and the structures seen initially rather than trying to figure out how they correlate to the 3D anatomy of the heart. For most of us it takes time to be able to connect the dots between what is seen on the screen and how we have to orient the probe in anatomic thoracic space. For that reason follows a somewhat extensive explanation of what protocol we use and how the probe marker should be oriented since learning how to orient yourself, and finding the correct marker direction, can be somewhat confusing in the beginning.

The correct way to orient the probe in this course as we, similar to the cardiologist, learn and teach the same orientation with the cardiac protocol, i.e. the marker on the screen should be on the right side as seen from the observer.
[Image credit: pocusbasics.org. ]
The image above is a reminder of how the probe marker is oriented when using the phased array probe and a cardiac exam protocol (with the marker on the screen pointing towards the right side of the screen). Note: in the beginning this is the most confusing part for novice POCUS practitioners as emergency physicians and cardiologists in USA use different orientations due to different traditions when doing bedside ultrasound of the heart (a fact you should bear in mind when watching any cardiac ultrasound videos on youtube done by American emergency physicians).
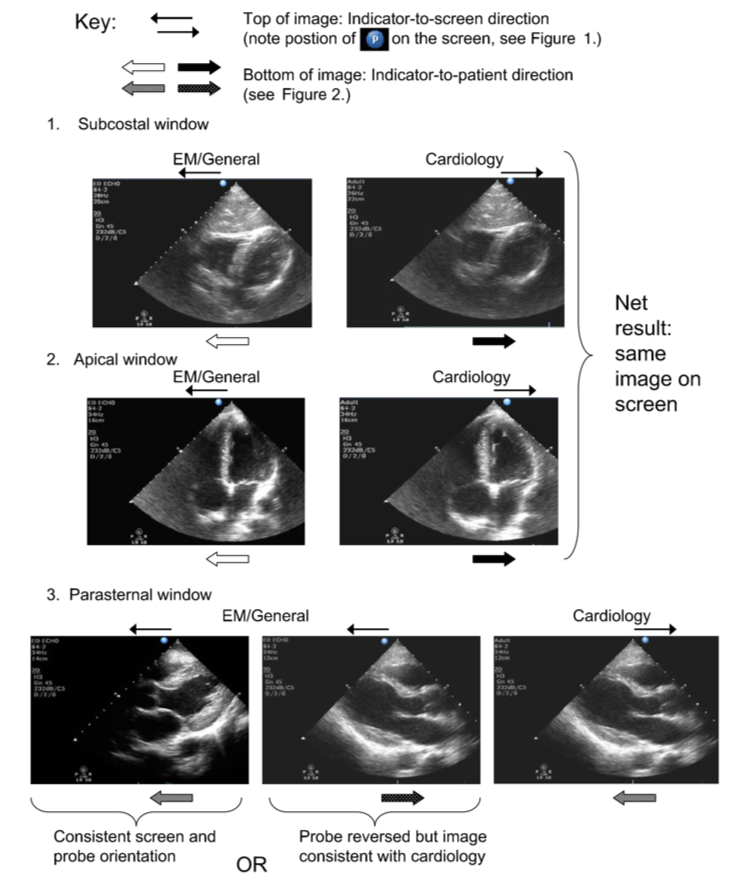
[Image credit: Dawson & Mallin, ‘Introduction to Bedside Ultrasound: Volume 1’, 2012.]
In short, the net result seen on screen is mostly the same except for the parasternal long axis (PLAX) view, where emergency medicine / general medicine the image is (sometimes) reversed by 180 degrees in certain clinics. For the benefit of having a general and common way of looking at the heart in the same way as the cardiologist we have adopted in our emergency clinics in Stockholm the same method of acquiring the images as the cardiologist’s and the biomedical examiners that perform echocardiography (see the clock image above for the correct probe marker and screen marker orientation in this course).
3. Subcostal (SC)
Subcostal video (5 min):
NOTE: the video above does not use a cardiac exam protocol, ie the marker on the screen is on the left left of the screen as opposed to the cardiac protocol where the marker is on the right and therfore, in this course, the marker on the probe should be aimed towards the patiens left (at 3 o’clock) instead.
[Credit: Phillips Perera & SonoSite]
We have already covered this view in the FAST module. The difference here is that we are using a cardiac protocol with the marker on the right side of the screen which means the same image is now acquired by putting the phased array probe with the marker towards the patients left side.
Cardiac arrest and the subcostal view
The subcostal view is a window that is also the most available view in cardiac arrest situations, since the chest area where PLAX/PSAX is obtained can be unavailable due to chest compressions (either by hand or a LUCAS machine). Since high quality compressions (and early access to a defibrillator) is the most important treatment when it comes to cardiac arrests we also want to, to the extend it is possible, avoid interrupting compressions or risk compromising the quality of compressions that can happen if we get ultrasound gel on the central chest area where they are performed. In most cases the subcostal view is enough to answer the two main question we want answered in a cardiac arrest situation:
- Is there pericardial fluid present?
- Is the heart beating?
In a recent systematic review by Kedan et al from 2020 *, when looking at 15 studies, the authors found that 14 of the studies showed a significant correlation between the presence of cardiac motion on ultrasound and short-term survival (most evident in patients with VF/VT). However as the authors point out; ‘currently there is insufficient evidence to recommend abandoning resuscitative efforts based on the absence of cardiac motion alone’. Having said that, the authors also note that the presence of cardiac motion is a ‘binary and simple assessment’ and ‘ultrasound can contribute valuable clinical applications in predicting short-term outcomes’. As always, point-of-care ultrasound adds a piece to the clinical assessment, along with arterial blood gas metrics, time to CPR and current length of CPR duration, cardiac rhythm, and other clinical information needed to help evaluate each individual case of cardiac arrest. Finally, and most importantly, as the authors point out:
‘While these results suggest POCUS provides additional diagnostic and prognostic information in the management of cardiac arrest, with the current body of knowledge regarding POCUS in the setting of cardiac arrest, we believe it remains vitally important that point-of-care imaging does not interfere with standard advanced cardiac life support efforts. With further study and perhaps a randomized multicenter trial using POCUS during treatment of cardiac arrest, perhaps standardized POCUS data can be incorporated in the evidence-based treatment of patients suffering from cardiac arrest. Point-of-care assessment of cardiac motion has the potential to be informative as an additive clinical data point in the clinical assessment of patients suffering from cardiac arrest.’ (emphasis in bold by me)
4. ParaSternal Long Axis (PLAX)
PLAX video part 1 (5 min):
PLAX video part 2 (5 min):
NOTE: the videos above does not use a cardiac exam protocol, ie the marker on the screen is on the left side of the screen as opposed to the cardiac protocol where the marker is on the right and thus the marker on the probe should be aimed towards the patiens right shoulder (at 10 o’clock) instead of the patiens left hip.
[Credit: Phillips Perera & SonoSite. ]
The PLAX view is (usually) obtained by putting the probe to the right of the sternum in a 90 degree angle on the patients chest with the marker aimed towards the patients right shoulder. In this view you should be able to visualize the left ventricle, the left ventricle outflow tract (LVOT), the mitral valve, the aorta valve, the left atrium, and part of the right ventricle. You should also be able to view the descending aorta, visualized as a circular structure just below the left ventricle/atrium in this view. The descending aorta is an anatomic landmark that can help to differentiate between pericardial and plural effusion, depending if the fluid is anterior (pericardial) or posterior (pleural) to the descending aorta.
5. Parasternal Short Axis (PSAX)
PSAX video part (4 min):
NOTE: the video above does not use a cardiac exam protocol, ie the marker on the screen is on the left side of the screen as opposed to the cardiac protocol where the marker is on the right and thus the marker on the probe should be aimed towards the patiens left shoulder (at 2 o’clock) instead of the patiens right hip.
[Credit: Phillips Perera & SonoSite.]
PSAX is obtained by tilting the probe 90 degrees from the position of PLAX until you have the left ventricle in a circular view, the marker should end up aimed somewhere towards the patients left shoulder. In this view you should, depending on the fanning-angle, be able to visualize the mitral valve in the middle. If you fan the prove towards the base of the heart (with the tail going down towards the stretcher) you should be able to visualize the aorta valve in the center, and the tricuspid to the left side of the screen (see image below). If you instead fan the probe towards the apex (with the tail moving away from the stretcher), moving the probe direction beyond the mitral valve, you should be able to visualize more proximal parts of the left ventricle including the chordae.

PSAX view with the probe fanned towards the base of the heart visualizing the aorta valve. The Mercedes Benz logo to the left of the image is a reminder to why the aorta valve is sometimes called the ‘Mercedes Benz sign’, as its common tricuspid configuration resembles the logo.
[Image credit: Renal Fellow Network ]
D-sign & Right Ventricle Strain

[Image credit: Konstantinides et al. ‘2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS): The Task Force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC)’. 2019. *]
In the PSAX view, at the level of chordae, when you fan the probe slightly more apical past the mitral valve, the left ventricle should be round/circular. If you notice that the septum is flattened, creating a “D-sign” looking shape, it could be a result from an enlarged right ventricle pressing against the left ventricle (see “C” in the image above). Right ventricle strain can have many causes, one of them being acute pulmonary embolism.
Not that the “chordae-view” is the main PSAX view and where assessments of right-ventricular strain or regional wall motion abnormalities in left ventricular radial movements should be done according to guidelines. The PSAX view of the mitral valve or base of the heart with the aortic valve present are additional cardiac windows to the four basic FATE windows.
Regional Wall Motion Abnormalities

[Image credit: Jacob Avila & 5 min sono, in the middle PSAX at the chordae-view with corresponding vascularizations of heart segments. 1* 2*]
PSAX (at chordea) is an excellent cardiac window for assessment of hypokinesis and regional wall motion abnormalities. In a healthy heart the structure should be symmetrically circular with symmetric movement and symmetric wall thickening in every part of the circle with each systolic cycle. For an excellent summary of bedside ultrasound and assessment of wall motion abnormality watch the following 12 minute video from westernsono.
EF can also be roughly estimated in this window, as a left ventricle area that decrease with approximately 50% with each cycle indicates a normal (left ventricle) heart function.
6. Apical Four Chamber View (A4C)
A4C video (5 min):
NOTE: the video above does not use a cardiac exam protocol as we will, ie the marker on the screen is on the left side of the screen as opposed to the cardiac protocol where the marker is on the right and thus the marker on the probe should be aimed towards the patiens left (at 3 o’clock) instead of the patients right.
[Credit: Phillips Perera & SonoSite]
The A4C view is obtained by aiming the probe below the patients left side with the marker aimed toward the patients left, and the probe slightly angled upward towards the patients thorax. This can be one of the more difficult views to get as you’ll often need to navigate in between ribs, and this view to can be easier to get if it’s possible to put the patients on her/his left side (the same applies to PLAX/PSAX as these views often are better depicted with the patient lying on their left side). In this view the four chambers should be visualized and by fanning the probe anteriorly a “five-chamber” view can sometimes be visualized, with the aorta in the middle. The five chamber view will be significant later as we provide one option to evaluate aortic stenosis in a later section in this module.
Right vs Left Ventricle and Right Ventricle Strain
A dilated right ventricle with basal RV/LV ratio >1.0 in the A4C view could be indicative of right ventricle strain. In this view it might be difficult to separate acute from chronic strain, however the assessment of right ventricle free wall for hypertrophy might help with differentiation.
Apical Wink / McConnell’s Sign & Right Ventricle Strain
McConnell’s sign, sometimes also called Apical Wink, is described as hypo- or akinesis of the right ventricular free wall with preservation of the apex. Initially McConnell’s sign was considered to be pathognomonic for acute PE *, however more recent studies have questioned that conclusion, and found that the sign and acute PE are not synonymous *. McConnell’s sign should be considered another characteristic of right ventricle strain and not a definitive diagnosis of PE.
7. IVC
IVC video (1 min):
[Credit: Ultrasound Critical Care.]
In POCUS the primary utility of learning to visualize the IVC has been to help aid physicians in assessment of the intravascular volume status of the patient. The reasoning has been that changes in volume status should be reflected in sonographic evaluation of the IVC, where increased (volume depletion) or decreased (volume overload) collapsibility of the vessel will help guide clinical management of the patient *. It has also been suggested in the literature that a measurement of >17-21 mm (at the hepatic veins, 1-2 cm from the RA) with less than 50% IVC respiratory collapsibility should indicate volume overload (or at least increased RA pressure) *.
However, as the intensivist and ECMO specialist dr. Chris Nickson at LITFL, points out:
“‘IVC is not a great test for intravascular volume status, fluid responsiveness or even fluid tolerance. Then again, it might be. The problem is that no-one really knows. A lot of big statements have come out of a lot of teeny studies that don’t really back them up.’ It’s probably better for ventilated patients than spontaneously breathing patients (you can remove much of the breath-to-breath variation that confounds so many of these assessment tools). It’s also probably OK at extremes (flat versus full) and serial measurements are probably a good idea.” *
Beside the validity problem with assessing the IVC, there is also a problem with reliability with bedside ultrasound as there is no consensus where/how to measure the IVC *.
Having said that, we have included IVC bedside sonography in this course as it is still practiced in many emergency departments (including our own), but for new POCUS practitioners it should be evaluated with caution (and, as always, in tandem to the bigger clinical context of each specific case). IVC sonography might not prove to be as promising for evaluating volume status as previously thought and further studies are needed. For now, these are the main points to keep in mind for IVC sonography (sampled/composed from LITFL * and ACEP *):
- Be a doctor, clinical context is more important than IVC ultrasound
- IVC probably does help at extremes (fat & full vs flat & collapsing), however:
- A plethoric IVC may occur in settings other than intravascular volume overload, such as cardiac tamponade, mitral regurgitation, or aortic stenosis (again, remember to consider clinical scenario)
- Intubated patients receiving positive pressure ventilation will have a reversal of IVC changes with respiration – the IVC diameter will be maximal with inspiration and minimal with expiration
8. TAPSE
TAPSE video (first 2.5 min):
[Credit: Jacob Avila & 5 min sono]
TAPSE is short for Tricuspid Annular Plane Systolic Excursion. This measurement is a parameter of global RV function. TAPSE correlates closely with the RVEF *, and has been found to be highly specific and relatively easy to measure *. We have included TAPSE in this course due to its relative simplicity and the fact that it can often be attained even if the image obtained is of less optimal quality.
You measure TAPSE at the A4C view of the heart by placing the 2D cursor at the tricuspid lateral annulus (in M-mode), and measuring the distance of systolic annular RV excursion along a longitudinal line defining the end of systole as the end of the T wave in the electrocardiogram. A TAPSE value of <17 mm indicates right ventricular dysfunction, while a TAPSE of < 14 mm indicates a poor prognosis in patients with chronic heart failure. A rule of thumb is that a high value when measuring TAPSE generally indicates no significant global function loss, while lower values require confirmation by a standard echocardiography. As with every other tool, bedside ultrasound is a part of the bedside clinical assessment and other adjuncts and tests should be included when necessary.
Visual estimation of TAPSE in A4C view
A recent study by Duanmu et al from 2020, found that a wide range of emergency clinicians demonstrated fair accuracy when estimating TAPSE visually (without M-mode & measuring) on prerecorded A4C clips after viewing a 5 minute educational clip *. The 70 emergency medicine participants viewed 20 prerecorded clips of A4C views and estimated if TAPSE was normal (>1.9 cm), borderline (1.5-1.9 cm) or abnormal (<1.5 cm) and compared their estimates to M-Mode measurements of TAPSE. The study found a pooled sensitivity of 88,6% and specificity of 81,6% for visual assessment of TAPSE, suggesting that visual assessment can be fairly accurate without extensive practice.
9. MAPSE
MAPSE video (2.5 min):
[Credit: theICUdoc]
MAPSE is short for Mitral Annular Plane Systolic Excursion. MAPSE is, similar to TAPSE, a parameter for assessing global function, but for the LV instead.
You measure MAPSE at the A4C by placing the cursor either on the septal- or lateral mitral annulus (in M-mode), and measuring the distance of systolic annular LV excursion along a longitudinal line defining the end of systole as the end of the T wave in the electrocardiogram. You could also measure both sites and then deriving a median metric from the two measurements (MAPSE can actually be measured from four sites of the atrioventricular plane corresponding to the septal, lateral, anterior, and posterior walls — for this course, the curriculum only includes learning to measure it from two of the sites).
A MAPSE ≥ 10mm indicates a preserved ejection fraction, while <8mm indicates impaired and <7mm severely impaired left ventricular function and in one study from 2012 * these cutoffs where associated with an LVEF of <50% and <30% respectively.
10. LVEF (Left Ventricular Ejection Function)
The fastest method for new POCUS practitioners to estimate LVEF is by doing a visual assessment. This can be done in all four views, but the preferred views for most are often the A4C and parasternal views, with ACEP (American College of Emergency Physicians) recommending an assessment of at least two cardiac windows * when estimating LVEF. By looking on the visualized walls of the left ventricle and estimating whether or not they move symmetrically and vigorously toward the center of the chamber during systole, and if they thicken as the muscle contracts, POCUS practitioners can roughly separate their estimation into three categories:
- hyperdynamic or normal (LVEF greater than 55%)
- moderate dysfunction (LVEF 30%-50%)
- severe dysfunction (LVEF less than 30%)
As mentioned, this is the fastest and most practical method for estimating LVEF but, perhaps, not the easiest as it can be difficult to assess left ventricular wall motion for beginners, especially in moderate cases, without first practicing for hours under proctored learning. That is why this module first introduced one relatively simple method of (indirect) measurement of LVEF with MAPSE.
There are of course other simple ways to measure LVEF such as EPSS (Mitral valve E-point septal separation) and not so simple ways such as the M-mode dimensional or Teichholtz method *.
We recommend students of this course to keep practicing estimating LVEF at the bedside, especially in patients known to have normal or depressed cardiac function, as we believe, in accordance with ACEP, that is the best way to become more proficient in visual estimation of LVEF.
11. Valve Diagnostics
Aorta Stenosis & Sclerosis
Aorta Stenosis & Sclerosis video (10 min):
[Credit: Katie Wiskar & westernsono.]
Even though valve-diagnostics is beyond the scope of this POCUS course, we’ve decided to include one method for evaluating aorta stenosis. The reason behind this is several:
- For students to gain some familiarity with the method and with applying doppler imaging
- Understanding the method improves knowledge and helps us interpret and understand accredited echosonography answers better
- In a longer perspective the evolution of POCUS will push the boundaries of bedside ultrasound, and we’d like this course to be a discussion forum for just that
- Several promising studies have shown physicians can develop specific echocardiography skills (like estimating left ventricular function *, pericardial fluid *, and assessment of basic hemodynamics *) and although valve-diagnostics has previously been considered outside the scope of POCUS, that too could change with time
For evaluating the aorta we’ll start at the PLAX and PSAX view in regular 2D view for a rough visual assessment: i.e. is the aorta leaflets thin and gracile or thick and more hyperechoic than usual (indicating calcification)? Then we’ll continue to A4C view fanning the probe slightly until an apical five-chamber view is visible. From the five-chamber view, continues wave Doppler (CW) will be applied passing through the aortic valve, measuring peak velocity. It is important to try to minimise the angle between the transaorctic flow and the the continuous wave Doppler. A spectral doppler tracing is applied to get a VTI envelope as demonstrated in the video. The VTI tracing will result in important numbers showing you the maximum velocity (VMax in cm/s) and mean pressure gradient (in mmHg). As the video points out in patients with sinus rhythm three measurements are recommended and the mean value used, and five measurements for patients with arrhythmia.

[Image credit: Messika-Zeitoun & Lloyd, ‘Aortic valve stenosis: evaluation and management of patients with discordant grading’, 2018. *]
The cut off between aortic stenosis and aortic sclerosis is a VMax of 2,5 m/s, anything above that threshold indicating aortic stenosis (see the image above). As mentioned in the video, patients with low cardiac output can give false low values due to severely reduced myocardial contractility that won’t be able to generate the pressure gradient that actively reflects the severity of the AS.
As usual, this exam should not replace other clinical adjuncts, and a professional echocardiography should be performed when necessary. However, as an echocardiography is not always available, this exam might help clinical management in bedside assessment of critically ill patients. And, although the data/studies for the accuracy of physicians performing bedside ultrasound for the evaluation of aortic stenosis and the training needed are rare and few, the study referenced in the video above showed some promising results *.
12. Pitfalls
- PLAX & pericardial vs pleural effusion: pericardial effusion will track between descending thoracic aorta and left atrium, pleural effusion will not.
- If you’re having difficulties finding the heart try placing the patient in left lateral decubitus position, which will bring the heart closer to the chest wall.
- Lung artifact is common and asking the patient to exhale can improve your image.
- Ribs may be obstructing the view, try moving the probe up and down, and in between, then angle the transducer towards the heart once you have a good acoustic window.
- Non-orthogonal planes can be mistaken for an enlarged right heart or D-sign. Make sure the LV looks round in the PSAX and that the apex is at the top of the screen in the apical windows.
- In a critically ill patient in the emergency room, do not struggle too long with trying to obtain an image if pressed on time, try instead shifting to another cardiac window
13. Putting It All Together
Following the FATE steps:
1. Look for obvious pathology
What jumps out in the cardiac image acquired? Is there a lot of pericardial effusion? PLAX is a good window to start with as it is a high yield ultrasound view that provides a lot of information. Does the dimensions of the right-ventricle, aorta, and/or left atrium seem off? Is any of them bigger than they should be, like the aorta; indicating type-A aneurysm? Does the mitral valve seem thin and gracile or is there a large blob on it, following along with it, with each heart beat, indicating endocarditis? Does it look like the mitral valve is barely opening – a sign of cardiac failure?
2. Assess wall thickness + chamber dimensions
Look for symmetrical wall thickness in the chambers during systole. Is the relationship between left and right ventricular as it should be, or does the right chamber seem bigger than usual? Look at other cardiac windows to help your assessment, do several windows support the pathology suspected?
3. Assess bi-ventricular function
PLAX and PSAX (at the chordae view) are excellent windows for assessing left ventricular function. Is the mitral valve hitting the septal wall in PLAX indicating a normal EF (see also EPSS). For right ventricle assessment look in A4C view, do a visual assessment of TAPSE: does the lateral annular part of the tricuspidal valve move up and down as it should? Measure TAPSE when unsure. Also assess wall thickness as mentioned above in step 2.
4. Image pleura on both sides
It takes an additional minute to complete a bedside cardiac ultrasound exam with a RUQ & LUQ view assessing for spine sign. Is there large amounts of pleural effusion on either/both sides due to the cardiac failure?
5. Relate the information to the clinical context
Does the findings match the clinical context? If not re-assess, do additional work-up, or ask for help interpreting your ultrasound images, from a more ultrasound experienced colleague.
6. Apply additional ultrasound
Do a complete lung ultrasound exam looking for B-lines or pleural-changes if you are differentiating between cardiac failure and pneumonia. Look at the abdominal aorta too if the aorta is widened in the parasternal long axis view and if you suspect an aneurysm. Look for a DVT (if you have the time and the patient is stable!), if you suspect right ventricular strain and pulmonary emboli. Etc.
The 9th, and last, question in the QUIZ below is a case-example of how to use the steps above from the FATE-protocol and how to put it all together in a clinical setting/case.
Summary & Disclaimer
For this module the focus is being able to obtain images of the four cardiac view and identify relevant structures in each image. Bedside cardiac ultrasound is the most difficult of the different exams when it comes to learning to orient the probe and visualizing decent quality images, which is why the focus is mostly on acquiring images during this course and only two focused questions:
- Is there pericardial fluid present?
- Is the heart beating?
The FATE questions relating to heart function with wall fall thickening and and bi-ventricular function, and dimension, and other obvious pathologies such as valve function, should be considered introductory and further practice beyond this course will be required for proficiency.
Keep in mind that the heart is a 3D structure and acquiring more cardiac windows when possible will help support your assessment of any pathology you suspect might be present.
We have taken cautions, as you might have noticed, not to make any big claims when it comes to our own ability or the ability of participating students to be able to assess cardiac function after this 5-day course. The reason for this is that reaching an acceptable proficiency threshold in basic bedside cardiac ultrasound for the secondary questions has a higher ceiling than the other modules/exams mentioned in this course. However, as some of the studies above suggest, and as one systematic review and meta-analysis by Jenkins et al published this year, even if the specificity and sensitivity when predicting LVEF, wall motion abnormality, and LV dilation and LV hypertrophy was greater for experienced than non-experienced operators, they still found that non-experienced operators were ‘able to detect cardiac abnormalities with reasonable sensitivity and specificity’ *. The problem is the heterogenecity of the the many studies they looked at, varying to a large degree in results, and in the absence of clear data for the amount of practice a “non-experienced” operator had.
Disclaimer:
Bedside cardiac ultrasound is difficult where small angles of the probe can have a large impact on how the heart is visualized which can make assessment of the structures and the function of the structures difficult or, worse, incorrect. Having said that, it can be a useful tool for bedside clinical assessment when used responsibly and will, over time, help the POCUS practitioner make better clinical judgements.
14. QUIZ
15. Further Reading
- BOOKS
- Noble. ‘Manual of Emergency and Critical Care Ultrasound’. 2nd Edition. 2011.
- Ch. 3 Cardiac Ultrasound
- Ch. 14 Ultrasound in shock
- pages 258-261 (IVC)
- Dawson & Mallin. ‘Introduction to Bedside Ultrasound – Vol 1’. 2013.
- Ch. 2. Basic Cardiac
- Dawson & Mallin. ‘Introduction to Bedside Ultrasound – Vol 2’. 2013.
- Ch. 1 Echocardiographic Assessment of Cardiac Output and Ejection Fraction
- Ch. 2 Right Heart
- Ch. 3 Diastology
- Soni et al. ‘Point-of-Care-Ultrasound’. 2nd Edition. 2020
- Ch. 13 Heart
- Ch. 14 Cardiac Ultrasound Technique
- Ch. 15 Left Ventricular Function
- Ch. 16 Right Ventricular Function
- Ch. 17 Inferior Vena Cava
- Ch. 18 Pericardial Effusion
- Ch. 19 Valves
- Ch. 21 Hemodynamics
- Ch. 22 Hypotension and Shock
- Ch. 23 Cardiac Arrest
- Noble. ‘Manual of Emergency and Critical Care Ultrasound’. 2nd Edition. 2011.
- SITES
16. References
- FATE (Focus Assessed Transthoracic Echo) protocol PDF:
- https://criticalusg.files.wordpress.com/2020/01/fate-card.pdf
- Original FATE study by Jensen et al. ‘Transthoracic echocardiography for cardiopulmonary monitoring in intensive care.’ 2004.
- PSAX
- https://www.renalfellow.org/2019/07/22/focused-cardiac-ultrasound-for-the-nephrologist-the-parasternal-short-axis-view/
- EPSS (End-Point Septal Seperation):
- E-point septal separation: a bedside tool for emergency physician assessment of left ventricular ejection fraction (2014)
- Koncise page from Emcurios about assessing shock in PLAX/A4C view and IVC:
- IVC
- Finnerty et al. ‘Inferior Vena Cava Measurement with Ultrasound: What Is the Best View and Best Mode?’ 2017.
- TAPSE
- Duanmu et al. ‘Visual Estimation of Tricuspid Annular Plane Systolic Excursion by Emergency Medicine Clinicians’. 2020.
- Kaul et al. ‘Assessment of right ventricular function using two-dimensional echocardiography’. 1984.
- Lee et al. ‘Effects of the HeartMate II continuous-flow left ventricular assist device on right ventricular function’. 2010.
- Schmid et al. ‘Tricuspid annular plane systolic excursion (TAPSE) predicts poor outcome in patients undergoing acute pulmonary embolectomy’. 2015.
- MAPSE
- Bergenzaun et al. ‘Mitral annular plane systolic excursion (MAPSE) in shock: a valuable echocardiographic parameter in intensive care patients’. 2013.
- Hu et al. ‘Clinical implication of mitral annular plane systolic excursion for patients with cardiovascular disease’. 2013.
- LVEF
- ACEP: Bedside Ultrasound Assessment of Left Ventricular Function
- Assessment of LV systolic function – ICU sonography — Tutorial 5
- Regional Wall Motions Abnormalities
- 12 minute video from westersono:
- MISC
- Ammari et al. ‘Prognostic Value of Computed Tomography Versus Echocardiography Derived Right to Left Ventricular Diameter Ratio in Acute Pulmonary Embolism’. 2021.
- Arntfield et al. ‘Point of Care Cardiac Ultrasound Applications in the Emergency Department and Intensive Care Unit – A Review’. 2012.
- Cowie et al. ‘Evaluation of systolic murmurs using transthoracic echocardiography by anaesthetic trainees’. 2011.
- Dresden et al. ‘Right ventricular dilatation on bedside echocardiography performed by emergency physicians aids in the diagnosis of pulmonary embolism’. 2013.
- Farsi et al. ‘Focused cardiac ultrasound (FOCUS) by emergency medicine residents in patients with suspected cardiovascular diseases’. 2017.
- Grapsa et al. ‘Assessment of Right Ventricular Structure and Function in Pulmonary Hypertension’. 2011.
- Jenkins et al. ‘Diagnostic accuracy of handheld cardiac ultrasound device for assessment of left ventricular structure and function: systematic review and meta-analysis’. 2021.
- Kimura et al. ‘Usefulness of a hand-held ultrasound device for bedside examination of left ventricular function’. 2002.
- Kedan et al. ‘Prognostic value of point-of-care ultrasound during cardiac arrest: a systematic review’. 2020.
- Konstantinides et al. ‘2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS): The Task Force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC)’. 2019.
- McConnell et al. ‘Regional right ventricular dysfunction detected by echocardiography in acute pulmonary embolism’. 1996.
- Mediratta et al. ‘Echocardiographic Diagnosis of Acute Pulmonary Embolism in Patients with McConnell’s Sign’. 2016.
- Mohamed et al. ‘The basics of echocardiography’. 2010.
- Royse et al. ‘Point of care ultrasound for basic haemodynamic assessment: novice compared with an expert operator’. 2006.
- Spruney et al. ‘Use of a hand-carried ultrasound device by critical care physicians for the diagnosis of pericardial effusions, decreased cardiac function, and left ventricular enlargement in pediatric patients’. 2005.
- Torbicki. ‘Echocardiographic diagnosis of pulmonary embolism: a rise and fall of McConnell sign?’ 2005.
- Walsh et al. ‘McConnell’s Sign Is Not Specific for Pulmonary Embolism: Case Report and Review of the Literature.’ 2015.
[Version 2.0 — Last updated 2022-06-28 — Status: Active]
[Version 2.0, update 2022-06-28: text revised, added section “Regional Wall Motion Abnormalities” under 5. PSAX, added section “Putting It All Together” to 13. Summary, and changed QUIZ , added question 9 as a case-example for the FATE-evaluation-steps.]
